SeeGene Bio receives FDA 510(k) clearance for EASYMADE TI patient-specific titanium implants
Originally reported by e-science.co.kr
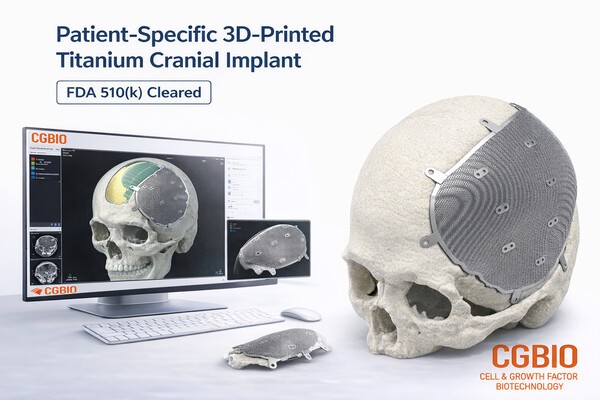
SeeGene Bio has secured US FDA 510(k) clearance for its EASYMADE TI, a line of patient-specific titanium implants designed for cranial and non-weight-bearing craniofacial reconstruction. The manufacturing process utilizes Laser Powder Bed Fusion (LPBF) to convert medical-grade titanium alloy powder into complex, anatomically precise structures based on patient CT data. CEO Yoo Hyun-seung confirmed that this regulatory milestone enables the company to enter the US market, leveraging a specialized design center to deliver finished products to clinical settings within a five-day turnaround window.
The approval addresses a critical gap in the reconstructive surgery market, where standard off-the-shelf implants often fail to match the unique anatomical contours of trauma or tumor resection sites. By utilizing LPBF, SeeGene Bio can implement porous lattice geometries that facilitate osseointegration, a significant advantage over traditional subtractive machining which lacks the design freedom for such micro-architectures. This move positions the company within the high-growth personalized medical device segment, competing against established players in the craniofacial reconstruction space by offering a vertically integrated digital workflow from CT scan to sterilized implant.
To succeed in the US healthcare system, SeeGene Bio must now demonstrate the reliability of its rapid five-day supply chain across diverse clinical environments. The company's ability to maintain tight tolerances and material integrity during high-speed LPBF production will be the primary factor in securing long-term contracts with US surgical centers. Future expansion will likely depend on the successful regulatory filing of additional implant geometries to broaden their reconstructive portfolio.
Topics