3D Systems launches NextDent Jetted Denture in Europe after Class IIa MDR certification, two months early
Hardware
Originally reported by 3D ADEPT
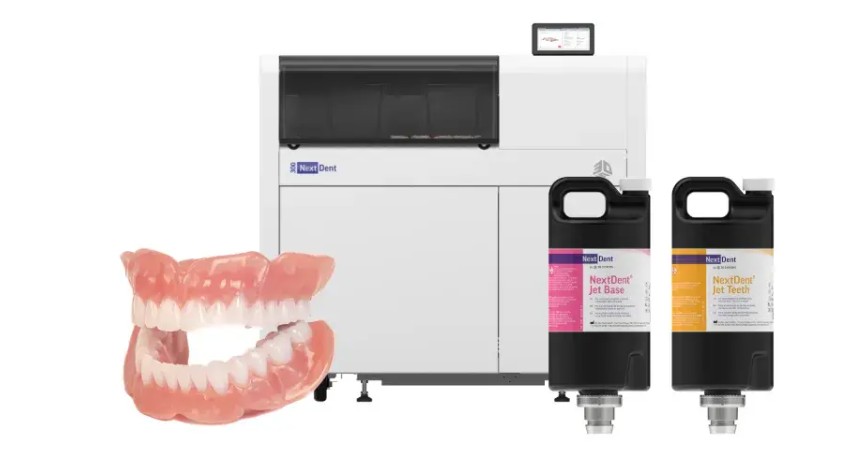
3D Systems has secured Class IIa certification under the EU Medical Device Regulation (MDR 2017/745) for its NextDent Jetted Denture Solution, enabling a full European commercial launch two months ahead of schedule. The certification covers the NextDent Jet Base and NextDent Jet Teeth materials, used with the NextDent 300 MultiJet printer, creating what the company calls the industry’s first monolithic, multi-material jetted denture workflow. This allows dental labs to produce patient-specific dentures in a single print, fully cured and ready for finishing without additional post-curing steps. The milestone builds on 3D Systems’ full-scope EU MDR certification announced in March 2026, demonstrating its ability to meet Europe’s stringent requirements for moderate-risk medical devices.
This move places 3D Systems squarely in the medical-dental vertical, a segment where regulatory compliance is the primary barrier to entry and the primary moat. The NextDent Jetted Denture solution directly competes with Stratasys’ PolyJet-based TrueDent workflow and desktop DLP systems from companies like SprintRay and Asiga, but 3D Systems differentiates by offering a complete, certified workflow—hardware, materials, software, and application expertise—from a single provider. The European dental lab market, estimated at several hundred million euros annually for AM-produced dentures, is fragmented but growing rapidly as labs shift from analog to digital workflows. By securing Class IIa certification ahead of schedule, 3D Systems gains a first-mover advantage in a market where switching costs are high once a lab embeds a certified workflow into its production line.
From an expert perspective, this is a textbook example of the IP lock-in grind pattern: 3D Systems has embedded its materials and printer into a certified workflow that dental labs will find difficult to replace without requalification. The practical next step for the company is to convert the early certification advantage into recurring material sales and service contracts, while competitors will need to match the regulatory bar before they can challenge this position. For buyers, the message is clear: if you want a turnkey, CE-marked denture production line today, 3D Systems is the only option in Europe.
Topics