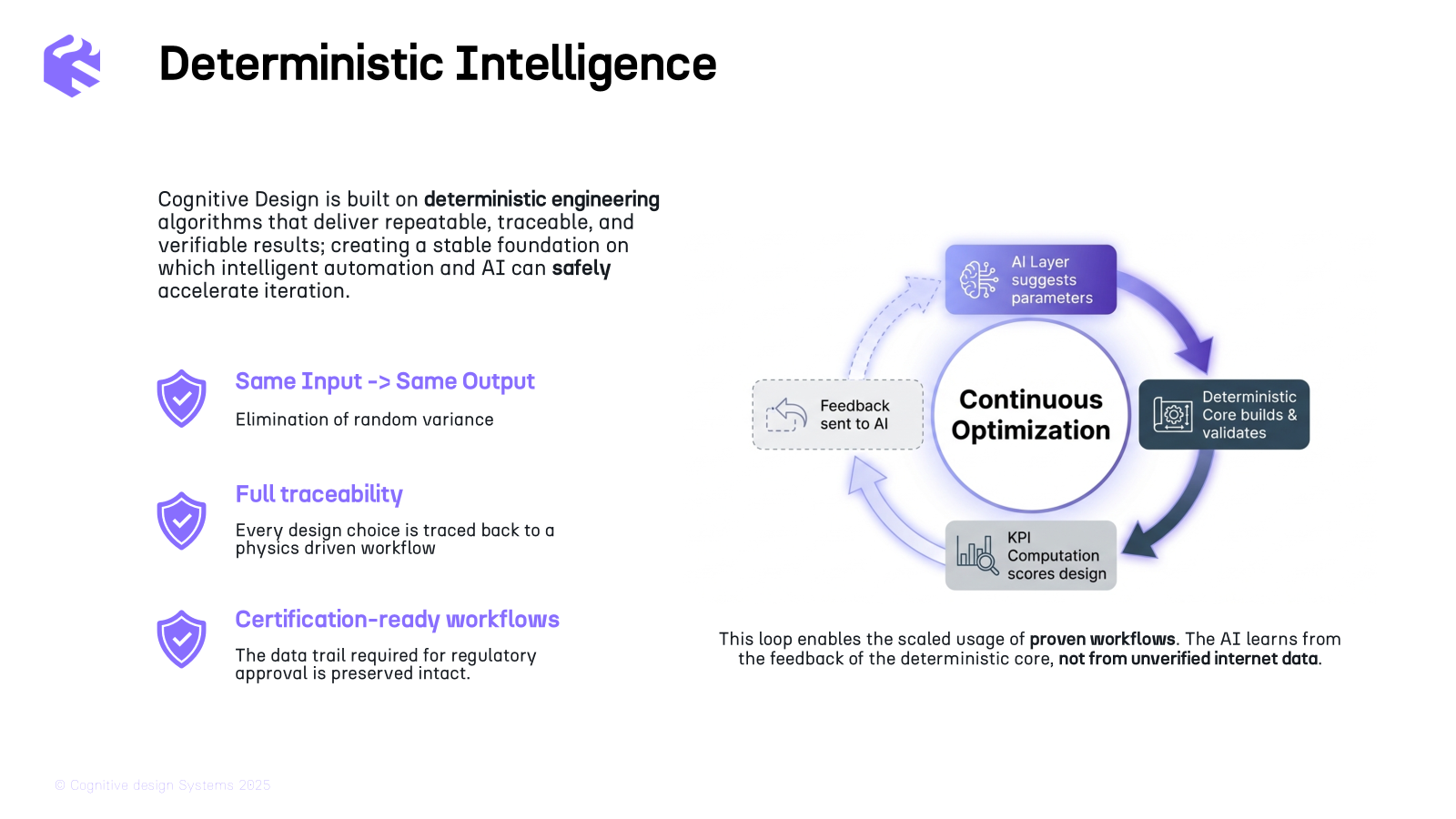
Technology
1 min read
Curiteva has received FDA nanotechnology designation for its Inspire 3D Printed Trabecular PEEK impl...
Curiteva has received FDA nanotechnology designation for its Inspire 3D Printed Trabecular PEEK implants. 🦴 Its HAFUSE technology features a 10-20 nm thick nanocrystalline layer that turns bio-inert PEEK into a hydrophilic, regenerative surface. By integrating 91.5 nm long nanocrystals into a 3D printed lattice, Curiteva achieves osseointegration once thought impossible for polymers. This marks a shift toward smart implants where AM dictates biological response at the molecular scale. 🚀 #AdditiveManufacturing #MedTech #3DPrinting #Curiteva